 Every time a patient swallows a pill, an intricate scientific journey begins—one that determines whether the drug will work, how quickly, and how safely. This journey, from oral administration to the site of action, is governed by two interdependent sciences: pharmacokinetics (PK) and pharmacodynamics (PD).
Every time a patient swallows a pill, an intricate scientific journey begins—one that determines whether the drug will work, how quickly, and how safely. This journey, from oral administration to the site of action, is governed by two interdependent sciences: pharmacokinetics (PK) and pharmacodynamics (PD).
Understanding this path is critical for pharmaceutical scientists, formulators, regulatory experts, and clinical developers alike.
Why Focus on Oral Administration?
Oral delivery remains the most common and preferred route of drug administration due to its convenience, non-invasiveness, and high patient compliance. However, it’s also the route with the most biological barriers—and where PK/PD considerations are essential to success.
What Happens After You Swallow a Tablet?
- Disintegration and Dissolution
- The drug formulation must disintegrate in the gastrointestinal (GI) tract and dissolve into solution.
- Formulation scientists optimize this stage using excipients, coatings, and solubility enhancers.
- Absorption
- The dissolved drug is absorbed across the intestinal lining, mainly in the small intestine.
- Factors influencing absorption:
- Solubility and permeability (classified in the BCS – Biopharmaceutics Classification System)
- pH-dependent ionization
- Food effects
- Transporters (e.g., P-glycoprotein efflux)
Pharmacokinetic impact: The rate and extent of absorption determine bioavailability—a key measure of how much active drug enters systemic circulation.
- First-Pass Metabolism
- Once absorbed, the drug enters the hepatic portal system and passes through the liver, where it may be metabolized before reaching the bloodstream.
- CYP450 enzymes in the liver can significantly reduce the active concentration of the drug.
Example: Propranolol has extensive first-pass metabolism, requiring higher oral doses than IV administration.
- Distribution to Target Tissues
- After systemic absorption, the drug must travel through the bloodstream to its intended site of action (e.g., brain, lungs, joints).
- Factors affecting distribution:
- Plasma protein binding
- Lipid solubility
- Blood–brain barrier permeability
- Volume of distribution (Vd)
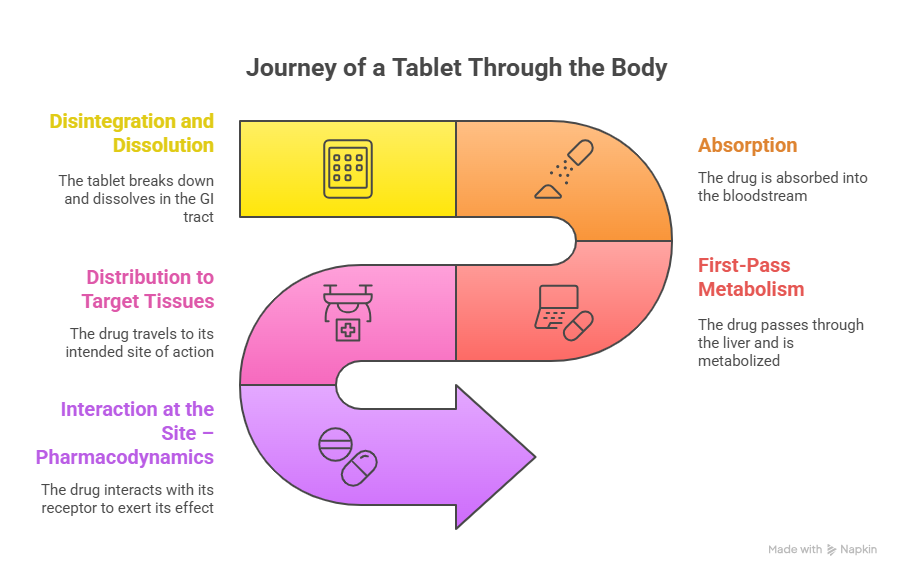
- Interaction at the Site – Pharmacodynamics
- Once at the target tissue, the drug must interact with its receptor or biological target to exert its therapeutic effect.
- The strength and duration of this interaction define the pharmacodynamic response:
- Onset of action
- Peak effect
- Duration of action
- Therapeutic window
Example: NSAIDs inhibit COX enzymes at sites of inflammation. While the drug reaches systemic circulation, PD dictates whether it achieves sufficient local inhibition.
Why Ongoing Training Matters
As regulatory expectations rise and new modeling tools emerge, continuous professional training in PK/PD is becoming essential.
Whether you’re in clinical development, regulatory strategy, formulation, or medical affairs, staying current with PK/PD methodologies, modeling software, and regulatory trends is key to making informed, data-driven decisions.
Investing in specialized courses and workshops helps teams:
- Interpret complex data with confidence
- Design more effective and efficient studies
- Communicate findings clearly to regulators and stakeholders
- Anticipate and mitigate development risks early
Leading pharmaceutical companies increasingly upskill cross-functional teams in PK/PD to bridge communication gaps between disciplines and accelerate drug development timelines.
Find out all about our upcoming professional PK-PD course <add link>
Integrating PK/PD: A Dynamic Relationship
The pharmacokinetic profile of a drug determines how much of it reaches the site of action, while pharmacodynamics tells us what it does once it’s there.
This interplay is essential for:
- Dosing regimen optimization
- Drug efficacy predictions
- Toxicity avoidance
- Formulation selection (e.g., immediate-release vs. sustained-release)
Case in Point: Controlled Release Formulations
If a drug has a short half-life but needs prolonged action, PK/PD modeling can justify sustained-release tablets that deliver therapeutic levels over time—improving adherence and outcomes.
Regulatory Relevance
Regulatory agencies require comprehensive data on how orally administered drugs reach and act at their target sites:
FDA (U.S.)
- Requires bioavailability (BA) and bioequivalence (BE) studies for oral products
- Guidance on food effects, dissolution testing, and population PK modeling
EMA (Europe)
- Emphasizes in vitro–in vivo correlation (IVIVC) for modified-release products
- PK/PD modeling is critical for dose selection and pediatric extrapolation
Key Standards
- ICH M9: Biopharmaceutics Classification System–based Biowaivers
- ICH E4: Dose–Response Information
- ICH E6(R3): Integrated approach to PK/PD in clinical trials
Common Challenges with Oral Drugs
- Low solubility (BCS Class II drugs)
- pH-sensitive degradation in the stomach
- Efflux by intestinal transporters
- Inconsistent food effects
- High inter-patient variability in metabolism
These challenges can mask or reduce efficacy, increase side effects, or delay market approval—unless PK/PD insights are proactively applied.

Conclusion
The path from oral administration to the site of action is one of the most scientifically complex and critically important journeys in drug development. While swallowing a tablet may seem simple to the patient, the science behind ensuring that drug reaches the right place, at the right time, in the right amount—and produces the desired effect—is anything but.
Pharmacokinetics (PK) and pharmacodynamics (PD) are not isolated academic disciplines; they are strategic enablers that support informed decisions at every stage of pharmaceutical R&D, formulation, and commercialization.
Key Points to Consider
- Oral Delivery Brings Convenience—But Also Complexity
From dissolution in the GI tract to passage through metabolic barriers, oral drugs face multiple hurdles that directly affect their therapeutic potential. - PK/PD Drive Dose Selection and Therapeutic Window Definition
Without a solid understanding of how the drug is absorbed, distributed, and acts on its target, determining optimal dosing is a shot in the dark. - Formulation and Route Must Be Aligned with Target Exposure
Poor correlation between drug delivery and drug action leads to product failure, even if the active molecule is promising. - Regulatory Agencies Require Robust PK/PD Justification
Submissions that lack coherent pharmacokinetic and pharmacodynamic rationale may face delays, rejections, or restrictions in approved labeling. - Variability in Patients Requires Population-Level Insight
Real-world use involves variability in metabolism, adherence, diet, and comorbidities. PK/PD models help anticipate and manage these challenges.
Consider Professional Development Opportunities
If your organization works in drug development or regulatory affairs, consider enrolling in or offering:
- Introductory and advanced courses in PK/PD modeling
- Workshops on IVIVC, PBPK, and exposure–response analysis
- Case-based training on regulatory submissions involving PK/PD
- Cross-disciplinary seminars to align R&D, clinical, and regulatory perspectives
Final Thought
Bridging the gap between oral drug administration and the site of action is not merely about achieving systemic exposure—it’s about designing medicines that are predictable, patient-friendly, and therapeutically impactful.
By embedding PK/PD thinking into every phase—from molecule to market—pharmaceutical innovators can reduce development risk, improve clinical outcomes, and bring safer, more effective treatments to patients around the world.
The next time a pill works exactly as intended, it’s because a multidisciplinary team asked the right PK/PD questions—long before that pill ever reached a prescription pad.